Agent
Snake fungal disease (SFD; also known as ophidiomycosis) is an emerging infectious disease caused by the fungal pathogen Ophidiomyces ophiodiicola. As the name suggests, this infection is only known to affect snakes and until recently had only been recorded in wild snakes from North America. Ophidiomyces ophiodiicola and SFD have recently been confirmed in wild snakes in Europe including Great Britain.
Findings from genetic studies and fungal cultures indicate that the European strains of O. ophiodiicola affecting wild snakes are distinct from the strains known to infect snakes in the eastern USA.
Species affected
Ophidiomyces ophiodiicola has been historically documented in captive snakes from multiple countries, including Great Britain. However, SFD in wild snakes in the eastern and midwestern United States of America first drew the attention of wildlife conservationists and researchers in 2006. Since then, a wide range of wild snake species have been affected in the USA.
In the UK, SFD was first detected in 2015 in a wild grass snake (Natrix helvetica: originally reported as Natrix natrix, updated subsequent to taxonomic revision in 2018) with skin disease found in the East of England. The snake was observed to be lethargic and in poor body condition with signs of dehydration. It was taken into care but subsequently died. At post-mortem examination, approximately 10% of the scales on the underside of the body had small (1-5 mm diameter) brown lesions with an irregular surface. Microscopic examination confirmed that the skin lesions were associated with a fungal infection. These signs are consistent with SFD and fungal culture and genetic sequencing confirmed the agent to be O. ophiodiicola.
As far as we are aware, this was the first-time O. ophiodiicola and SFD had been found in a wild snake outside of North America.
Further testing of grass snake carcasses and skin sheds with lesions has detected O. ophiodiicola from multiple locations across England. Ophidiomyces ophiodiicola has also been detected in a dice snake (Natrix tessellata) from the Czech Republic and a grass snake (N. helvetica) from Switzerland (originally reported as N. natrix, updated subsequent to taxonomic revision in 2018).
Continued monitoring is required to determine if the other native snake species (i.e. European adder (Vipera berus) and smooth snake (Coronella austriaca)) in GB are susceptible to O. ophiodiicola infection and/or SFD.
Signs of disease
SFD in wild snakes in the USA has variable presentations, including thickened or ulcerated skin with scabs or crusty scales and abnormal moulting. Subcutaneous nodules (swellings under the skin) and swelling of the face and head can also occur. The fungal infection is typically limited to the skin and rarely invades the internal organs. Experimental work and anecdotal observations from the USA indicate that sick snakes with SFD may behave abnormally, spending increased amounts of time in exposed locations. They may also increase their frequency of moulting and become anorexic.
Whilst there is some evidence of wild snake population declines in the USA due to SFD, especially for already threatened populations and species, further work is required to establish the conservation significance of this disease in North America.
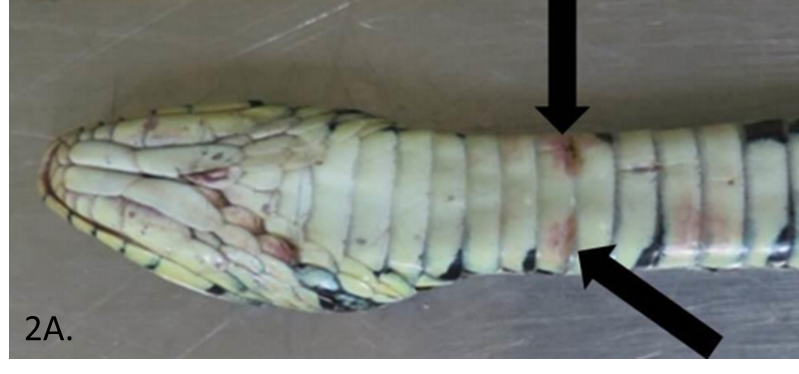
Our understanding of the significance of SFD in grass snakes in GB, and the potential for this fungus to infect other snake species, is currently limited. From the small number of cases examined to date, the skin lesions in grass snakes vary in severity and colour (pale to dark brown), can be mild (1-2mm in diameter) and are most commonly located on the underside of the head and body. Consequently, mild skin lesions are unlikely to be noticed in the field. A small number of post-mortem examinations have confirmed SFD in grass snakes in GB where the condition is considered to be severe and likely to have contributed to their death. The extent to which the skin lesions cause generalised ill health in grass snakes requires further investigation.
Grass snakes are widely distributed in lowland England and Wales, although they have undergone declines due to habitat loss. There are some records from Scotland but their status there requires investigation. Further research is underway to understand the significance of O. ophiodiicola to the health of wild grass snakes and to grass snake populations in GB.
Figure 1A. Eastern rat snake (Pantherophis alleghaniensis) from New Jersey, USA, showing signs of snake fungal disease. Obvious external abnormalities are an opaque infected eye (spectacle) and roughened, crusty scales on the snout. (Photograph by D.E. Green, USGS National Wildlife Health Center.)
Figure 1B. Eastern rat snake from New Jersey, USA, showing signs of snake fungal disease with crusting on lateral scales. (Photograph by D.E. Green, USGS National Wildlife Health Center.)
Figure 2A. Grass snake (Natrix helvetica) with skin lesions due to SFD (Black arrows). (Photo credit: Zoological Society of London.)
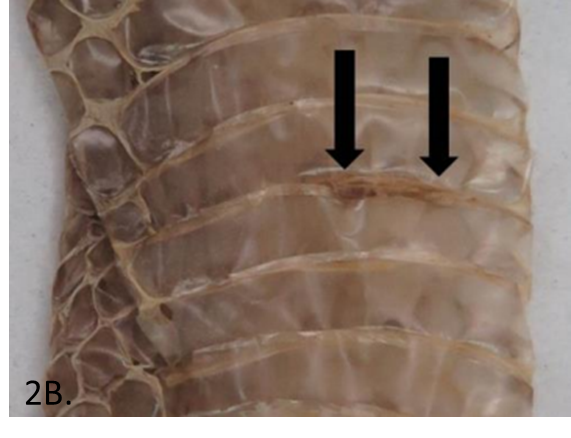
Figure 2B. Grass snake skin shed with lesions positive for Ophidiomyces ophiodiicola (Photo credit: Zoological Society of London.)
Disease transmission
Experimental studies have shown that O. ophiodiicola infects snakes when it comes in direct contact with their skin, and that infection might be facilitated by superficial scale damage. Studies of the fungus suggest that it may occur and survive in the environment (e.g. soil). The extent to which transmission occurs directly between snakes, or indirectly through the environment, is unknown.
Distribution and origin
Ophidiomyces ophiodiicola has been detected from wild snakes with a wide distribution across England since 2010, which suggests that this fungus is established in GB. There is no evidence to indicate that O. ophiodiicola was recently introduced to Europe from North America or vice versa. Instead, it is likely that it has been present for some time in GB but was not previously recognised.
In 2016, a single skin shed from a dice snake (Natrix tessellata) from the Czech Republic with lesions also tested positive for the fungus, confirming its presence in wild snakes in mainland Europe. In 2018, a grass snake (N. helvetica) from Switzerland also tested positive for O. ophiodiicola, with skin lesions suggesting it was suffering from SFD.
Health surveillance of wild reptiles is challenging due to their cryptic and often solitary nature. Surveillance across GB is required to gain better understanding of the distribution of infection. It is currently unknown if the strain of O. ophiodiicola detected is native to GB (i.e. has always been present) or has been introduced.
Similarly, continued surveillance is required to establish whether SFD is a missed endemic disease (i.e. is a regular occurrence at a stable level) or an emerging one (i.e. occurring at an increasing frequency) and what its impact, if any, is on snake populations in Great Britain.
Population genetic studies of O. ophiodiicola indicate that multiple recent introductions of the fungus have occurred into North America. Since the fungus has been detected in captive snakes in multiple countries, transcontinental movement of snakes could have led to its spread.
Risk to human health
No known risk to human health.
Risk to domestic animal health
Ophidiomyces ophiodiicola is only known to infect snakes. The fungal infection has been documented in captive snakes from multiple countries, including Great Britain. It is possible that O. ophiodiicola may be transmitted between wild and captive snakes. Herpetologists with captive snakes are recommended to maintain good biosecurity but since pet snakes are typically housed indoors within vivaria, the pathways for transmission are limited. Substrate from vivaria housing snakes should be not be discarded where wild snakes might gain access.
It is illegal to release non-native snake species to the wild in the UK under the Wildlife & Countryside Act 1981.
A Pet Code of Practice (link below) has been developed by the Ornamental Aquatic Trade Association (OATA) and the Reptile and Exotic Pet Trade Association (REPTA) to help pet owners understand their responsibilities. This Code is based on a Council of Europe document which has more detailed advice on keeping pets (link below).
Diagnosis
There are multiple potential causes of skin disease in wild snakes, therefore it is not possible to reach a diagnosis without specialist laboratory testing.
If you wish to report finding a dead wild reptile, or signs of ill health in wild reptiles, please visit www.gardenwildlifehealth.org. Alternatively, if you have further queries or have no internet access, please call the Garden Wildlife Health vets on 0207 449 6685.
We do not encourage members of the public to disturb or handle wild snakes.
If a sick or injured snake is observed, we recommend that a local veterinarian, the RSPCA or an experienced herpetologist or wildlife rehabilitator is contacted for advice.
We also appeal for snake skin shed samples (if near intact) and details of the date and location from which the shed was collected. If you have such samples, please store them separately and contact us via the Garden Wildlife Health website or call us.
Prevention and control
There are no effective measures known for the treatment or control of SFD in wild snakes.
Ecological consultants and herpetologists should consider biosecurity protocols as routine, such as wearing disposable gloves, cleaning and disinfection of equipment, especially storage buckets, boots and artificial refugia, particularly between sites. Special dedicated equipment should be used when working with wild snakes and must never be shared between captive and wild snakes. Wild snakes should not be brought into captivity in an environment where non-native snakes are kept, unless there is a sound justification and rigorous biosecurity measures are implemented. People keeping non-native snakes must adhere to biosecurity measures when undertaking fieldwork.
A disease risk analysis should be incorporated into the planning of wild-to-wild reptile translocations and disease risk management should be implemented.
Further reading
USGS National Wildlife Health Center: Snake Fungal Disease: Snake Fungal Disease | U.S. Geological Survey (usgs.gov)
A Pet Code of Practice has been developed by the Ornamental Aquatic Trade Association (OATA) and the Reptile and Exotic Pet Trade Association (REPTA) to help pet owners understand their responsibilities. This Code is based on a Council of Europe document which has more detailed advice on keeping pets: Pet-Code-of-Practice.pdf (ornamentalfish.org)
Scientific publications
Di Nicola MR, Coppari L, Notomista T, Marini D (2022) Ophidiomyces ophidiicola detection and infection: a global review on a potential threat to the world’s snake populations. European Journal of Wildlife Research 68(5) p.1-29 doi.org/10.1007/s10344-022-01612-8
Ladner JT, Palmer JM, Ettinger CL, Stajich JE, Farrell TM, Glorioso BM, Lawson B, Price SJ, Stengle AG, Grear DA, Lorch JM (2022) The population genetics of the causative agent of snake fungal disease indicate recent introductions to the USA. PLoS Biology 20(6):e3001676 doi.org/10.1371/journal.pbio.3001676
Meier G, Notomista T, Marini D, Ferri V (2018) First case of Snake Fungal Disease affecting a free-ranging Natrix natrix (Linnaeus, 1758) in Ticino Canton, Switzerland. Herpetology Notes 11:885–891. www.biotaxa.org/hn/article/view/37196
Franklinos LHV, Lorch JM, Bohuski E, Rodriguez-Ramos Fernandez J, Wright ON, Fitzpatrick L, Petrovan S, Durrant C, Lintom C, Baláž V, Cunningham AA, Lawson B (2017) Emerging fungal pathogen Ophiodiomyces ophiodiicola in wild European snakes. Scientific Reports 7:3844 doi.org/10.1038/s41598-017-03352-1
Lorch JM, Knowles S, Lankton JS, Michell K, Edwards JL, Kapfer JM, Staffen RA, Wild ER, Schmidt KZ, Ballman AE, Blodgett D, Farrell TM, Glorioso BM, Last LA, Price SJ, Schuler KL, Smith CE, Wellehan JFX, Blehert DS (2016) Snake fungal disease: an emerging threat to wild snakes. Philosophical Transactions of the Royal Society B 371: 20150457. doi.org/10.1038/s41598-017-03352-1
Acknowledgements
Current funding for the GWH comes in part from Defra, the Welsh Government and the Animal and Plant Agency (APHA) Diseases of Wildlife Scheme (DoWS) http://apha.defra.gov.uk/vet-gateway/surveillance/seg/wildlife.htm; and from the Esmée Fairbairn Foundation, the Universities Federation for Animal Welfare and the Garfield Weston Foundation.
Disclaimer
This fact sheet was produced by Garden Wildlife Health (GWH) for information purposes only. The GWH will not be liable for any loss, damage, cost or expense incurred in or arising by reason of any person relying on information in this fact sheet.
Date of factsheet update:
September 2022